
Chemists describe solutions in terms of concentration. We define the concentration of a solution as the amount of solute in a given volume of solution. - ppt download

MOLARITY A measurement of the concentration of a solution Molarity (M) is equal to the moles of solute (n) per liter of solution M = n / V = mol / L Calculate. - ppt download

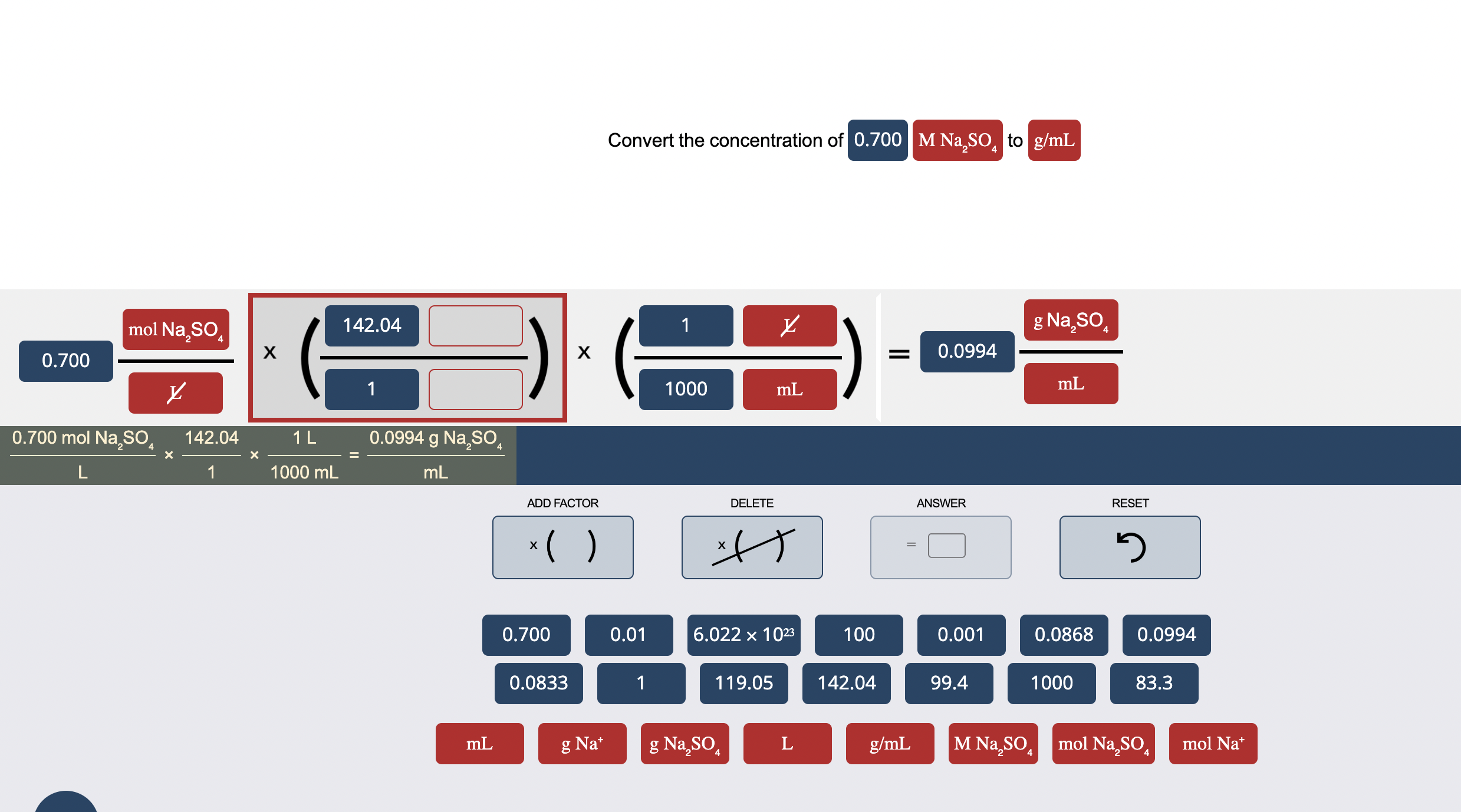















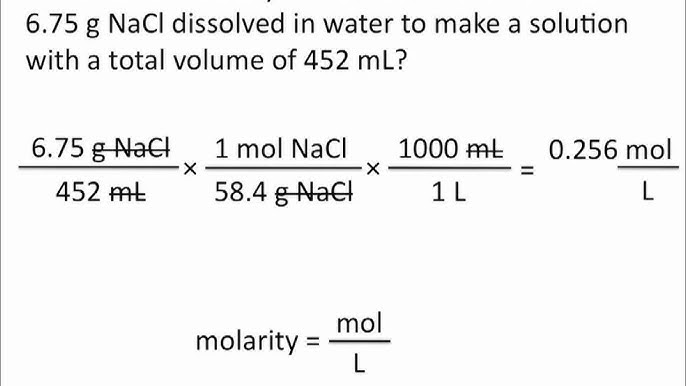

![Molarity Calculator [with Molar Formula] Molarity Calculator [with Molar Formula]](https://scrn-cdn.omnicalculator.com/chemistry/molarity@2.png)