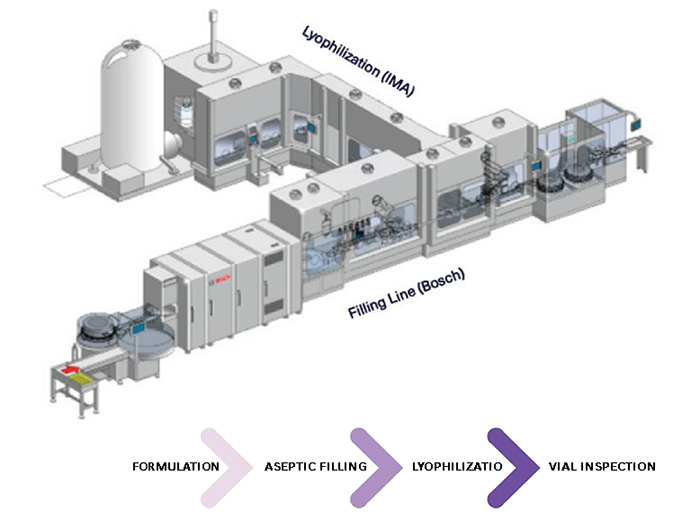
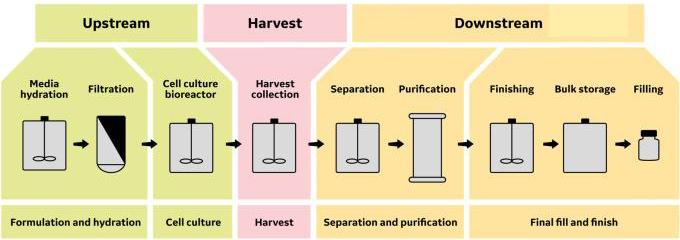
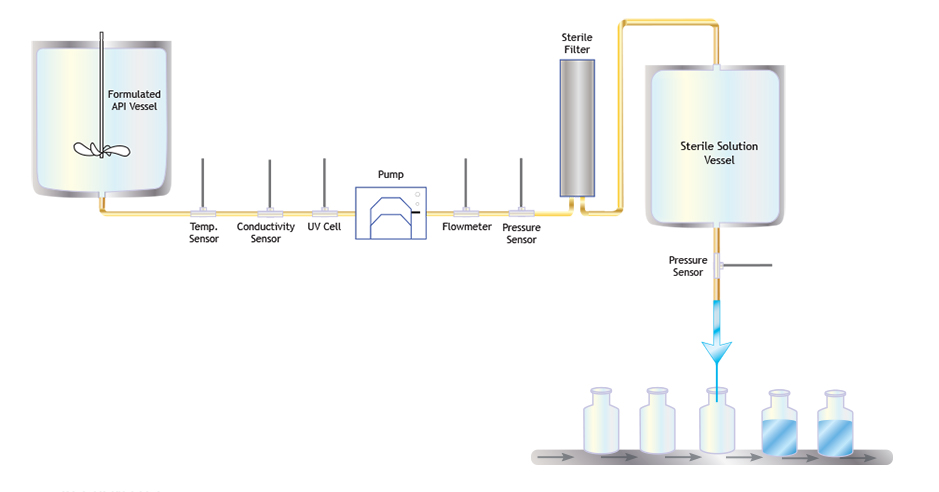
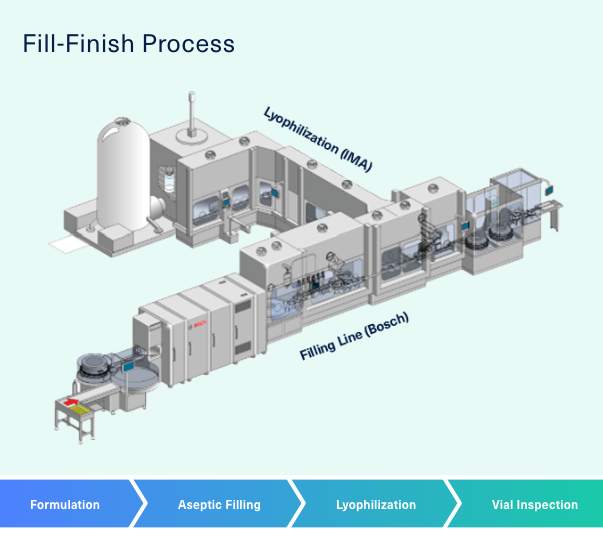
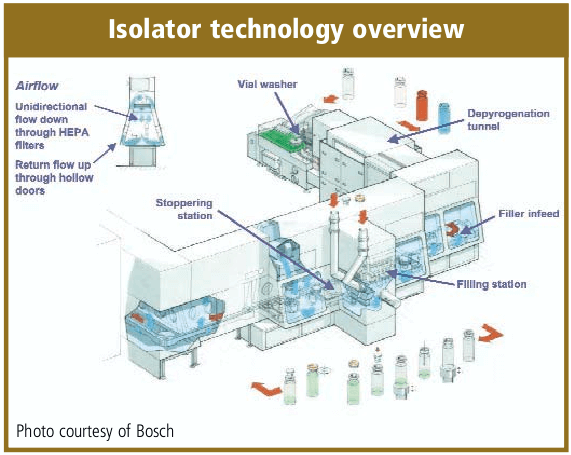
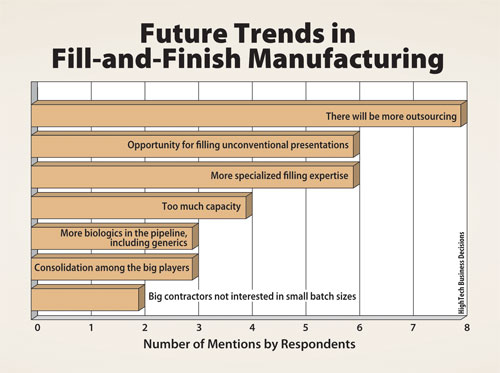
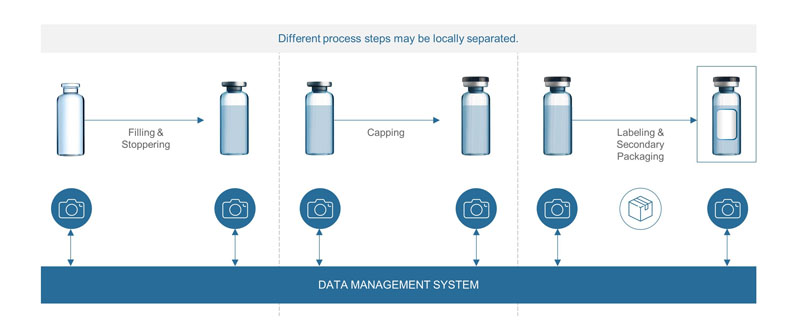
Automation of a Single-Use Final Bulk Filtration Step: Enhancing Operational Flexibility and Facilitating Compliant, Right–First-Time ManufacturingBioProcess International
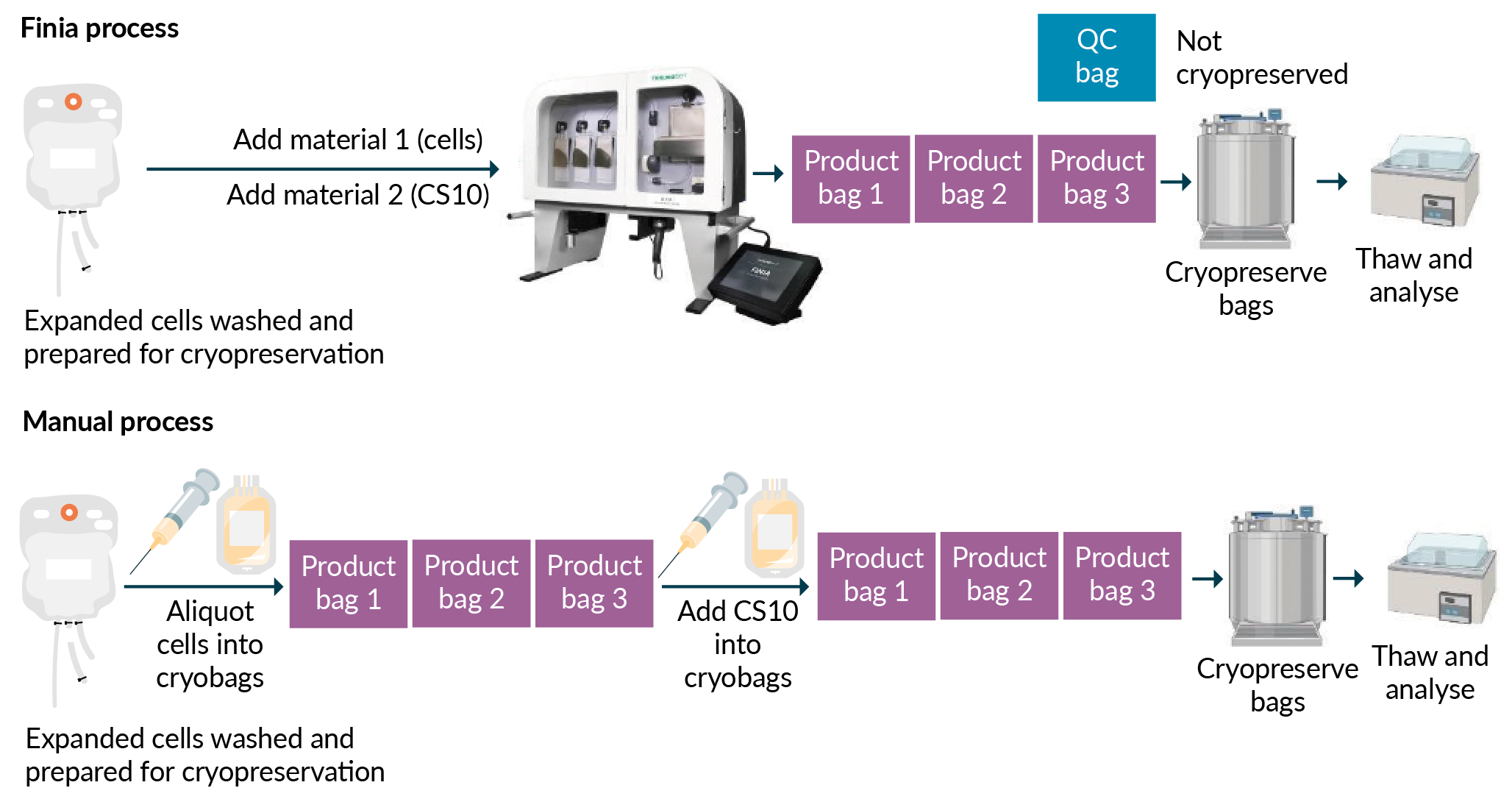
De-risking the final formulation, fill and finish step in cell therapy manufacturing: considerations for an automated solution

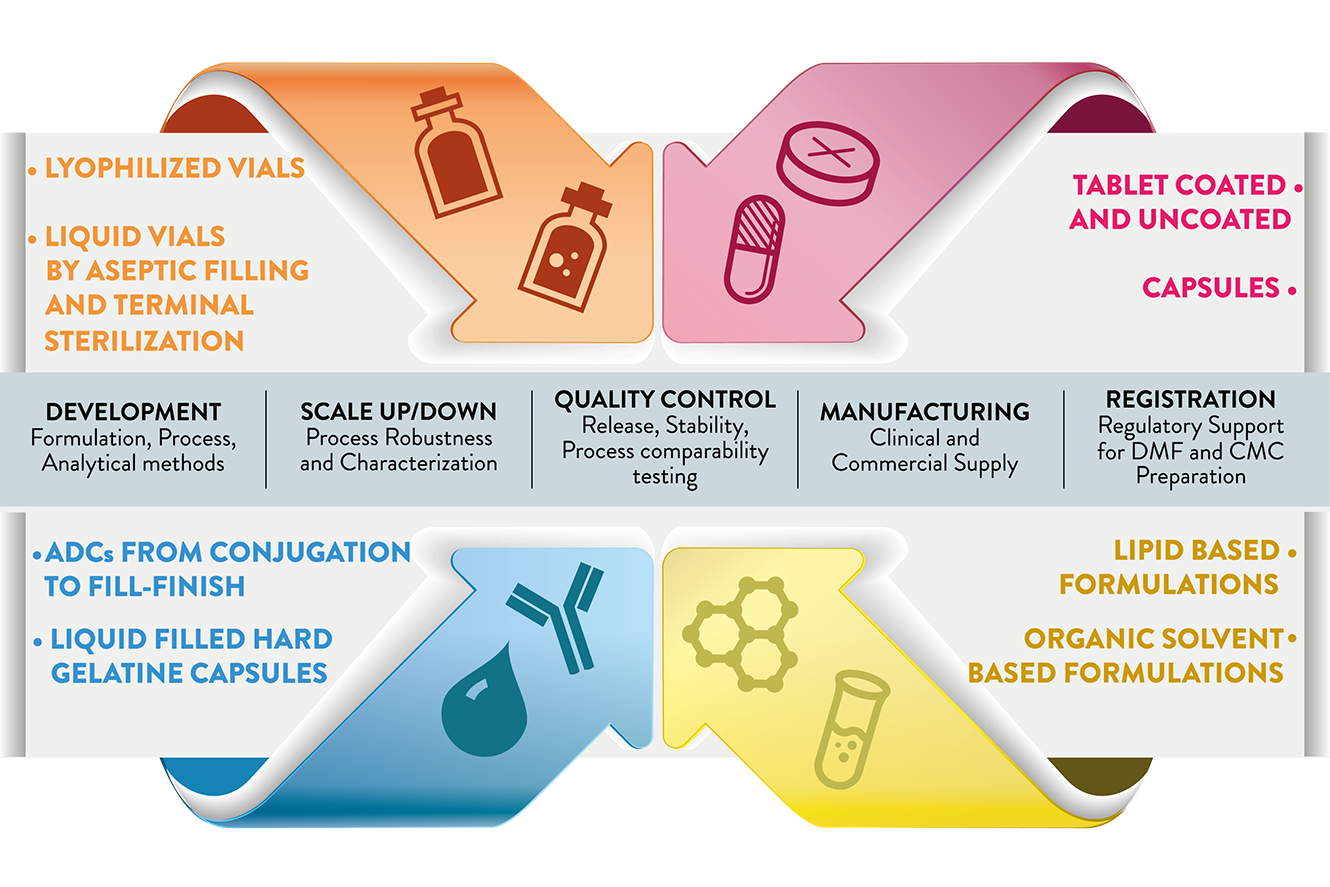
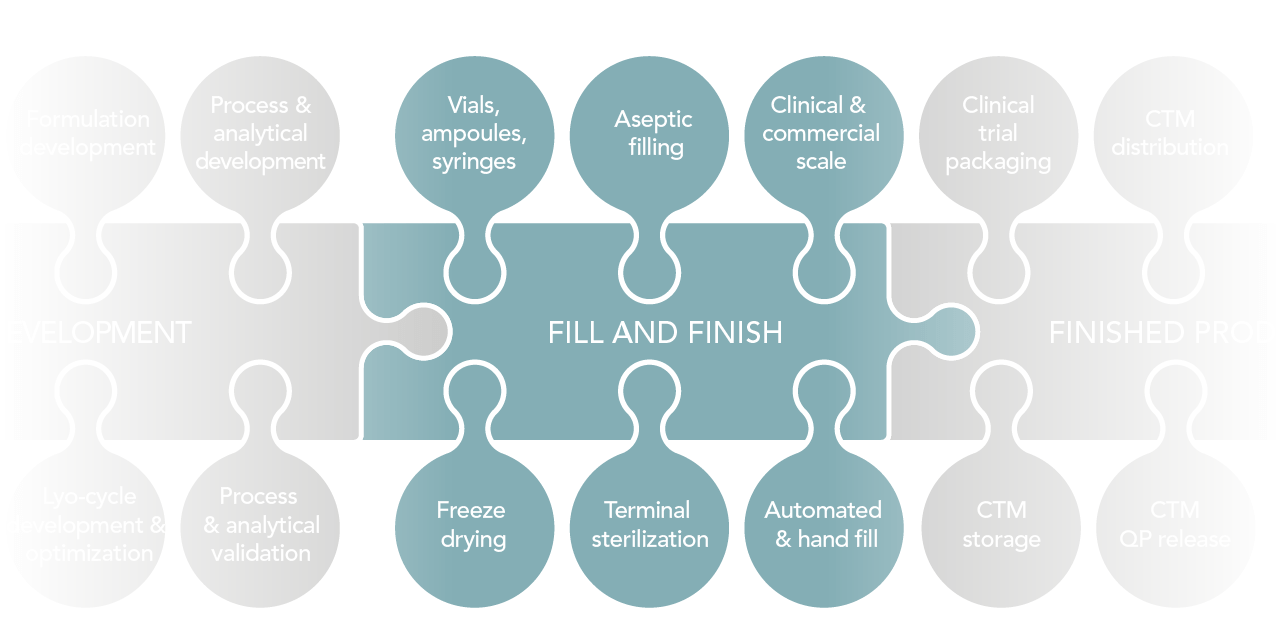
Critical Factors for Fill–Finish Manufacturing of Biologics - BioProcess InternationalBioProcess International